by Anne-Caroline Genix1, Vera Bocharova2, Bobby Carroll2, Philippe Dieudonné-George1, Michael Sztucki3, Ralf Schweins4, Alexei P. Sokolov5, and Julian Oberdisse1.
1 Laboratoire Charles Coulomb (L2C), Université de Montpellier, CNRS, F-34095 Montpellier, France
2 Chemical Sciences Division, Oak Ridge National Laboratory, Oak Ridge, TN 37831, USA
3 European Synchrotron Radiation Facility, 71 Avenue des Martyrs, CS 40220, F-38043 Grenoble Cedex 9, France
4 Institut Laue-Langevin, DS/LSS, 71 Avenue des Martyrs, CS 20156, F-38042 Grenoble Cedex 9, France
5 Department of Chemistry, University of Tennessee, Knoxville, TN 37996, USA
Polymer nanocomposites (PNCs) are materials usually made of nanoparticles immerged in a polymer matrix. They possess striking macroscopic properties which are related to their intriguing dynamical behavior on the macro- and nanoscale, and in particular at the interfaces between the nanoparticles and the polymer. In PNCs where the matrix is a mixture of molecules of different molecular weights, several unusual effects can only be explained if one assumes a peculiar spatial distribution of the short and long chains throughout the samples. [Bocharova et al, ACS Appl. Nano Mater. 2020, 3, 3427] This motivated the present study on where the different chains are actually located.
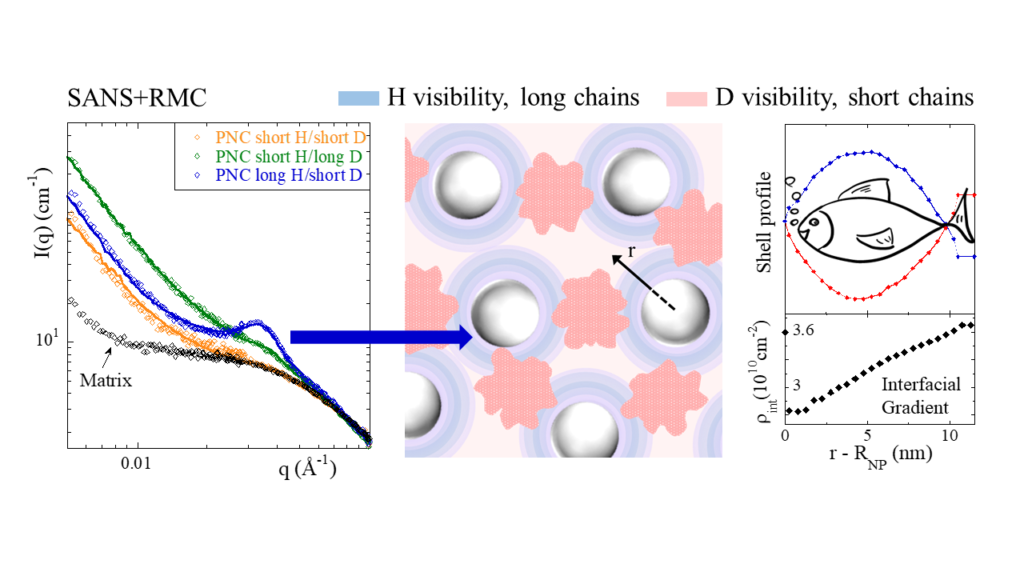
In this work, we address the question of concentration fluctuations of the short and long chains in PNCs using small-angle neutron scattering (SANS) and isotopic labeling in presence of contrast-matched nanoparticles (NPs), i.e., NPs are invisible to neutrons. The first surprising result of our study is that while the hydrogenated (H) and deuterated (D) poly(vinyl acetate) chains mix at a molecular level, they do present spatial fluctuations of two different types in PNCs with attractive polymer−silica interaction, beyond the random-phase approximation. A picture of simultaneous bulk and interfacial segregation of the polymer blend in presence of NPs is proposed based on specifically developed numerical evaluation methods of the scattered SANS intensity. The resulting spatial distribution is illustrated in the central figure, with bulk segregation in red, and interfacial effects in blue. The corresponding new large-scale organization strongly depends on chain mass asymmetry in the PNC blend. The silica is found to systematically induce bulk segregation. In addition, only with long H-chains, a strong scattering signature as shown by the peak of the blue curve on the left is observed in the q range of the NP size : it is the sign of interfacial isotopic enrichment, that is, of contrasted polymer shells close to the NP surface. The isotopic density profile within the shell has been characterized by reverse Monte Carlo simulations revealing the existence of a large interfacial layer (over ca. 10−20 nm depending on the NP size) with an unexpected shape, as shown on the right-hand side it looks like a fish ! The occurrence of two solutions – up and downside of the fish – results from the squared contrasts in the calculation of the scattered intensity.
Fundamental thermodynamic considerations provide some simple understanding of these puzzling observations of the influence of chain asymmetry. In all cases, NP surfaces trap the polymer blend in a non-equilibrium state. There seems to be an isotopic preference for the H-chains to adsorb, while the longer chains undergo an entropic driving force towards the interface. This results in preferential adsorption around NPs only if the chain length and isotopic preference toward the surface combine their entropic and enthalpic driving forces, i.e., for long H-chains.